Mimicking
Of Hydrothermal Processes Can Lead To Formation Of Porous Material
Scientists can make a covalent organic
framework (COF) material without using an organic solvent by boiling the water
in a closed cylinder far above its boiling point - a process they call geomimetic hydrothermal synthesis as it
mimics some of the harsh conditions deep beneath the crust of Earth.
COFs are created by combining organic building
components together using covalent bonds. COFs have several features which
make them the perfect material for electrochemical devices, gas storage,
and detectors, ranging from structural adaptability to high chemical stability.
Water has now been utilized in place of the organic solvents i.e. polyimide that were previously employed to create COFs. Organic solvents need careful handling and waste removal after use. Byeong-Su Kim of Yonsei University South Korea, who conducted the study, explains this entire phenomenon. Water, on contrary, is free of these drawbacks. It is not generally a suitable solvent since the majority of the organic molecules are not dissolved in it.
Kim's research team was intrigued by the notion that numerous intriguing chemical processes, such as the production of highly crystalline minerals, may occur deep below the Earth and wanted to mimic some of the such circumstances. 'We discovered that under hydrothermal conditions, ionic product of water and dielectric constant became identical to those of the organic solvents,' Kim adds. ‘Under harsh circumstances, the hydrogen bonding in water molecule begins to degrade.’
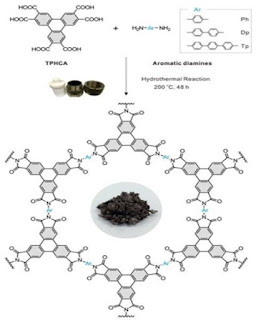
They created multiple solid
polyimide-based COFs after boiling(200°C) a solution comprising of the two precursors
i.e a diamine and a carboxylic acid in a closed flask for 2 days. Kim's research team discovered that when
polyimide COF was tested as a functional anode in batteries, it offers twice
more efficiency and it can also be recharged 200x more consistently than
graphite anodes used in typical lithium-ion batteries. The higher efficiency is
because of the large porous structure of COF, which improves the transport of
ions.
Rubén Mas Ballesté, a researcher at the
Autonomous University of Madrid in Spain, is less enthused. He emphasizes that
the lack of availability of building material, instead of the synthetic
technique, is what restricts COF output. As a result, implementing the study's
conclusion on an industrialized scale would be prohibitively expensive. Ballesté
adds that imide formation is widely known for COF preparation and that water
has already been recorded for the production of other COFs at lower
temperatures.
Currently, Kim and his coworkers are exploring
several water-soluble antecedents to build new COF structures.
About the Author: Eishah Khalid is a student
of BS Chemistry at The Islamia University of Bahawalpur, Pakistan.
.png)
0 Comments